
News, Insights, and more on Industrial IoT
 In our last blog post, we gave an overview of the four types of sensors most commonly used in wastewater treatment facilities. Now we want to zoom in and take a closer look at how sensors help us measure water quality in particular.
In our last blog post, we gave an overview of the four types of sensors most commonly used in wastewater treatment facilities. Now we want to zoom in and take a closer look at how sensors help us measure water quality in particular.
Maintaining water quality is crucial, of course, because nobody wants to drink, bathe, or use dirty fluids for...anything, really. However, there’s more to keeping tabs on water quality than meets the eye.
How you measure water quality at a wastewater treatment plant isn't just about the final product. Water quality needs to be maintained to support treatment processes, automate critical wastewater treatment processes, ensure proper solids processing, and minimize the impact of discharged effluent. Wastewater treatment plant controls use sensors to measure water quality so that these processes work as they should.
Now, when it comes to wastewater treatment, the following water quality characteristics are essential:
Sensors are key because they empower wastewater plant operators to collect rich data on each of these core variables. Below, we dig into the sensors used to measure these things and explain why these water quality sensors are so important.
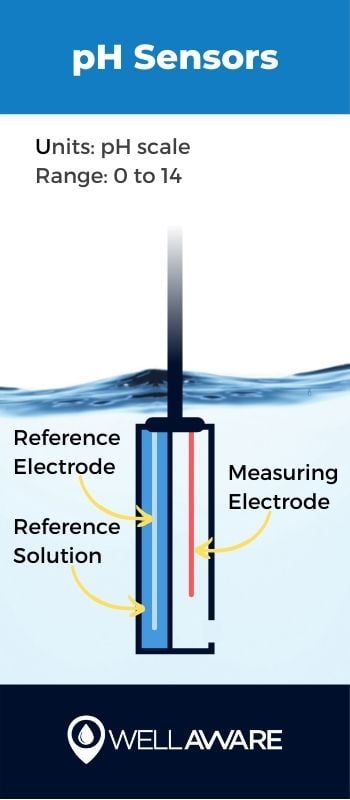
We describe water’s acidity or alkalinity using the pH scale. Acidic water has pH levels between 0 and 7, and alkaline (aka basic) water has pH levels between 7 and 14.
Water’s pH level often is important because it indicates its inherent aesthetic quality; water that is too acidic or alkaline may simply taste bad. For municipal drinking water supplies, water treatment plants usually target pH levels between 6.5 and 8.5. Beyond simple aesthetics, however, pH must be tracked to ensure treatment processes can take place effectively. Water pH can serve as a proxy for the general condition of water and indicate whether there are substances creating acidic or alkaline conditions that would affect treatment processes.
Sensors measure pH using electrodes submerged in water. Measuring electrodes compare electrical signals to a reference electrode, allowing the sensor to detect variations in pH.
pH scale
0 to 14
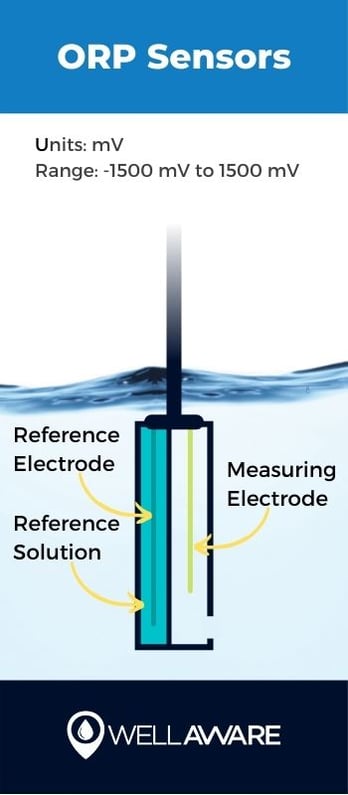
Oxidation-reduction potential (ORP) refers to water’s ability to transfer electrons through oxidation (i.e., giving electrons away) or reduction (i.e., taking on electrons). ORP is analogous to pH in that the measurement can reveal whether water is acidic or basic. In general, the more acidic water is, the more likely it is to accept electrons. The more alkaline, the more likely it is to give electrons.
ORP reveals if conditions are acceptable for certain treatment processes, for example, if various kinds of bacteria that aid in wastewater treatment can do their job effectively. Wastewater treatment plants use both bacteria and chemicals to condition water and remove substances like nitrogen and phosphorus, so if ORP conditions aren’t ideal, these processes will lose efficiency or become entirely ineffective.
Sensors calculate ORP by comparing measuring electrodes to reference electrodes. The mechanism is similar to how sensors measure pH levels.
Millivolts (mV)
-1500 to 1500 mV
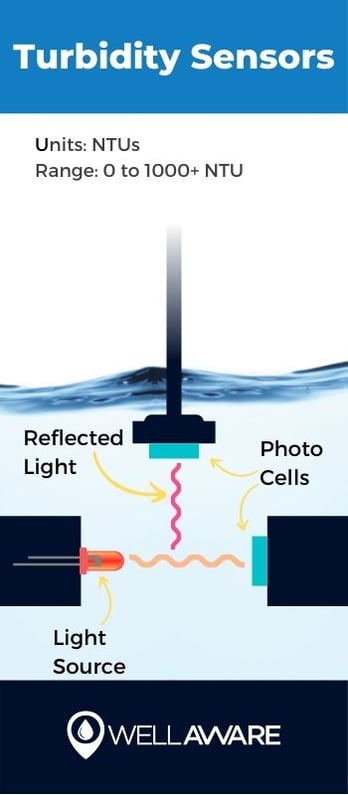
Turbidity is a measure of water clarity. It tells us how much light can pass through liquids. Clear water - the kind of water that people want to drink - lets more light pass through.
Operators use turbidity to evaluate not only the aesthetic quality of water but also the efficacy of disinfectants. Water with high turbidity impedes UV rays, which not only reduces UV disinfection efficiency but may also indicate the presence of harmful bacteria or other organic matter.
Sensors, often called nephelometers, measure turbidity by sending beams of light into liquids that are reflected back at a 90-degree angle. These light-detecting sensors pick up how much light gets reflected back - the more light that returns to the sensor (that is, the less light that passes through), the higher the turbidity.
Nephelometric Turbidity Units (NTUs)
0 to 1000+ NTU
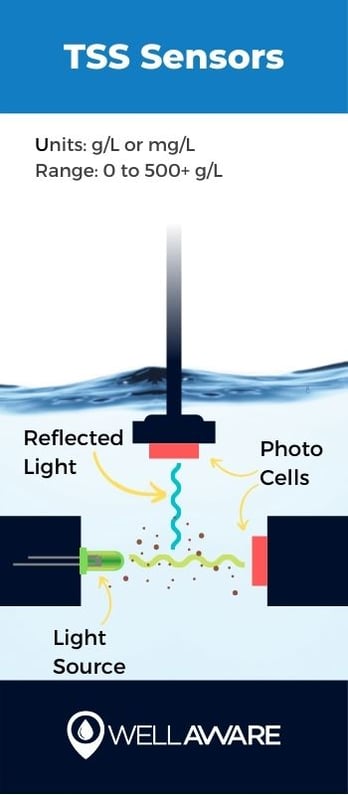
Total suspended solids (TSS) is a measure of the concentration of solids suspended in water (obviously). Solids in wastewater may include silt, sediment, and other particles, none of which people want to see come out of their faucets.
TSS is similar to turbidity, and the two are often correlated. TSS points to drinking quality and disinfection effectiveness. Water with a high concentration of suspended solids can indicate ineffective disinfection processes. Suspended solids can also affect the amount of oxygen in water and impact treatment.
TSS is typically measured using the same mechanism as turbidity sensors, that is using light detectors. In fact, many turbidity- and TSS-sensors are sold as a single combined sensor. However, TSS is measured using different units and ranges, indicating the mass of suspended solids per volume unit of water.
Typically milligrams per liter (mg/L) or grams per liter (g/L)
0 to 500+ g/L
Dissolved oxygen is a measure of how much gaseous oxygen molecules are dissolved in water.
Oxygen levels are a harbinger of water health. Water with too little oxygen often indicates high levels of pollution, whereas water with too much oxygen can inhibit anoxic treatment processes. In water treatment, we track two measures: chemical oxygen demand (COD) and biological oxygen demand (BOD) to determine how much dissolved oxygen must be present in the water for effective chemical and bacterial treatment. For instance, bacteria need “anoxic conditions” to remove nitrates - if there’s too much oxygen present, bacteria can’t do their job.
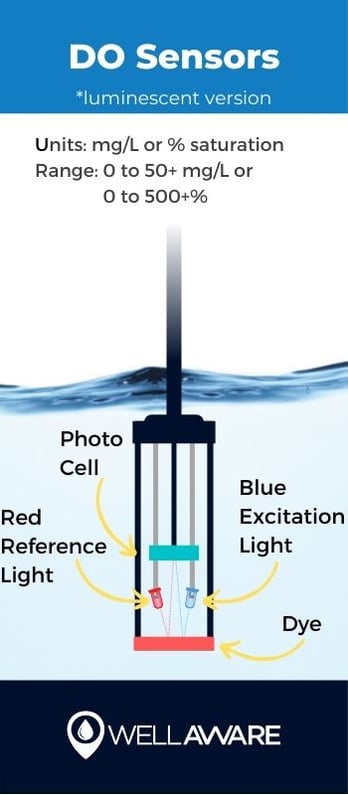
Dissolved oxygen can be measured in two different ways: using optical or electrochemical sensors. Optical sensors, often called luminescent DO sensors, use LEDs, a sensor film with dye, and light detectors to measure how much oxygen is present in water. Electrochemical sensors measure changes in electrical current caused by the presence of oxygen.
Typically mg/L or % saturation
0 to 50+ mg/L, 0 to 500+ % saturation
Note: There are conditions in which oxygen can become oversaturated in water, with a saturation percentage greater than 100%, which is why DO sensors will detect ranges above 100%.
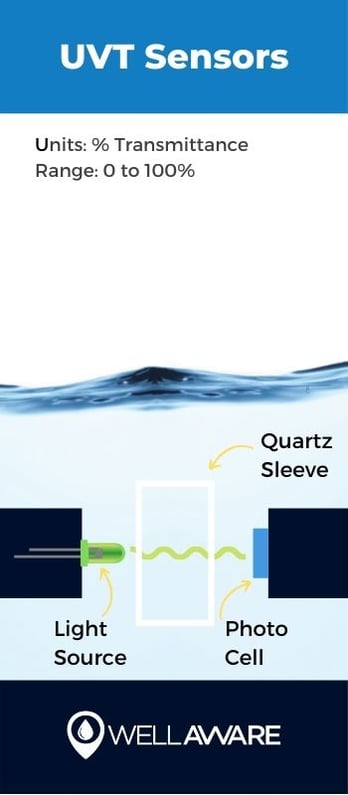
UVT sensors measure how much UV light is absorbed in water, and/or how much light is transmitted through the water, specifically at the 254 nm wavelength.
UV absorption measurements act as a corollary for how much organic matter is in water. Water treatment processes should remove all harmful organic matter before distributing it to municipal water supplies... because no one wants organic matter - like E. Coli and feces - coming out of their showerheads. UVT sensors tell us how much disinfection is required or how effective disinfection has already been.
UV transmittance sensors work by using UV light to shine through quartz cells that contain water samples. A light detector is placed opposite the generated beam to measure how much ultraviolet light is transmitted through the water, and/or how much was absorbed by organic matter.
UV transmittance (UVT) %
0 to 100% UVT
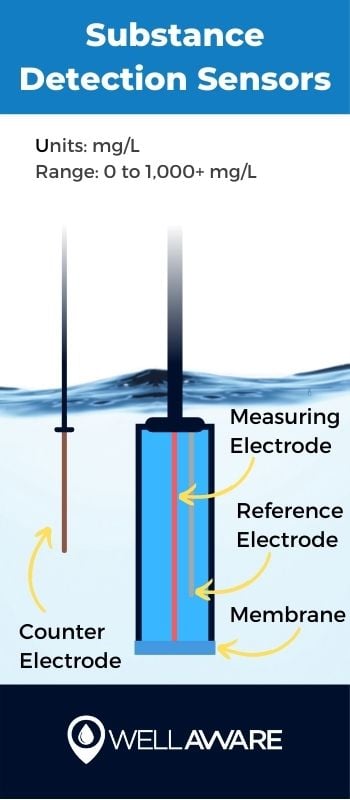
Substance detection refers to the process of using specialty sensors to detect various compounds or molecules in water streams.
Substance detection is key for identifying ammonium, nitrates, phosphates, potassium, fluorides, and chlorine, among other substances. These sensors, which vary in their detection specialties, provide important measures of water quality both before and after chemical and biological treatment processes.
Sensors use special electrodes - specific to the particular compound they are trying to detect - to measure changes in electrical properties, indicating the presence of said compounds.
mg/L
Typically 0 to 1,000+ mg/L
Measuring and maintaining water quality is only one piece of a much larger puzzle. Wastewater treatment is complex, which is why we’ll explore another group of sensors in our next post: process instrumentation sensors.
Until then, feel free to check out our Water & Wastewater Remote Monitoring web page for more information on how WellAware supports water treatment plants, utilities, and municipal water systems.
Like what you're reading? Sign up for updates!
Have a Question?